Abstract
This clinician‑facing review compares six commonly discussed "skin booster" domains: injectable polynucleotides (PN/PDRN/PN‑HPT), platelet‑rich plasma (PRP), topical retinoids (retinol/retinal/tretinoin), topical peptides, "exosome" preparations, and amino‑acid/HA biorevitalisers (Sunekos and Jalupro). "Skin boosters" is an umbrella term used inconsistently across markets; therefore, the analysis is structured around (a) biologic mechanism plausibility and (b) human clinical outcomes, durability, and safety.
Mechanistically, the modalities cluster into three families. First, transcriptional remodellers (retinoids) alter keratinocyte differentiation and dermal matrix turnover through nuclear receptor signalling (RAR/RXR‑mediated gene regulation). Second, autologous or biomaterial "signal" therapies (PRP; PN/PDRN) aim to re‑programme tissue repair responses. Third, biorevitaliser nutrient cocktails (Sunekos, Jalupro) target extracellular matrix support through HA hydration plus amino acid/peptide provision.
For practice, a pragmatic sequencing model emerges: use topical retinoids as first‑line "priming" for most photoageing phenotypes, then layer injectable modalities based on dominant concerns, while reserving higher‑variability interventions for patients with appropriate tolerance for uncertainty, cost, and protocol iteration.
Introduction and Scope
This article addresses skin "quality" interventions — hydration, texture, fine rhytids, mild laxity, dyschromia, and early dermal matrix decline — rather than volumisation (dermal fillers for contouring) or neuromodulation. Where commercial language uses "skin booster" to include HA‑based injectables and adjunctive biorevitalisers, each modality is evaluated on:
- Mechanistic plausibility at the molecular/cellular level (pathway mapping)
- Human clinical outcomes (study design hierarchy, objective measures, durability)
- Safety/adverse events and contraindications
- Practical protocols and sequencing (priming, induction, maintenance)
- Consistency and variability (biologic variability, product standardisation)
- Regulatory status and sourcing (CE/UK/US positioning and supply governance)
Search Approach
A pragmatic evidence strategy was used: peer‑reviewed literature (RCTs, split‑face trials, systematic reviews/meta‑analyses), manufacturer product sites for composition and device status claims, and regulatory/government sources including UK acceptance timelines for CE‑marked devices, EU cosmetics regulation, UK medicines/ATMP guidance, and US FDA warning letters.
Mechanisms of Action: Pathway Detail
Polynucleotides and PDRN
In aesthetic medicine, "polynucleotides" typically refers to purified fragments of DNA polymers (often from fish/salmonid sources) supplied as injectable gels or solutions. PDRN (polydeoxyribonucleotide) is a related term for mixtures of double‑stranded deoxyribonucleotide chains (chain length range 80–2,200 base pairs), commonly extracted and purified from salmon trout gonads.
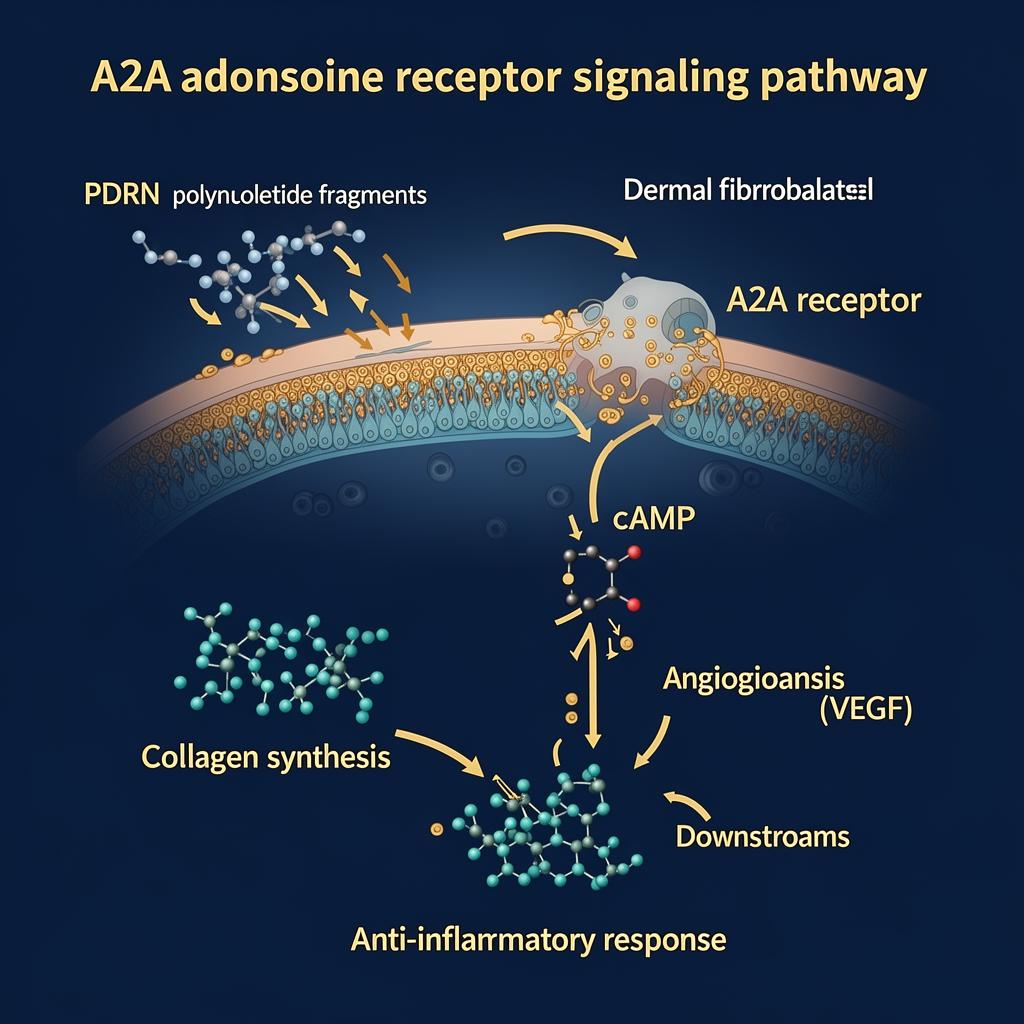
A2A Receptor Signalling
Adenosine signalling is mediated by P1 receptors (A1/A2A/A2B/A3). A2A and A2B receptors stimulate cAMP production, while A1/A3 inhibit adenylate cyclase. PDRN's anti‑inflammatory and regenerative effects are repeatedly linked to A2A receptor activation, aligning with decreased pro‑inflammatory cytokine expression and improved wound healing/angiogenesis in experimental models.
The "salvage pathway" concept: stressed, hypoxic, or aged tissues may have constrained de novo nucleotide synthesis; provision of nucleotide fragments can be recycled into nucleic acids, supporting repair processes. Fibroblasts internalise PDRN‑derived nucleotides and utilise them for salvage DNA synthesis.
PRP: Growth Factor Delivery and Immune‑Matrix Modulation
PRP is an autologous blood‑derived concentrate created by centrifugation to enrich platelets above baseline. Modern classification efforts (e.g., DEPA: dose, efficiency, purity, activation) were proposed specifically because biological differences in PRP preparations may underpin heterogeneous clinical outcomes. PRP is best conceptualised as a patient‑specific biologic whose performance depends on both patient physiology and preparation protocol.
Retinoids: RAR/RXR Transcriptional Control
Retinoids exert effects by binding nuclear retinoic acid receptors (RARα/β/γ) which function as transcription factors — classically as heterodimers with retinoid X receptors (RXR). Clinically used retinoids differ in "distance to retinoic acid": retinol and retinal require enzymatic conversion, whereas tretinoin is all‑trans retinoic acid itself. A 2025 clinical review summarises tretinoin's mechanisms as including regulation of epidermal differentiation, activation of fibroblasts, induction of collagen synthesis/recycling, inhibition of collagen loss, and reduction of matrix metalloproteinases.
Topical Peptides
Cosmeceutical peptide classes include signal peptides (matrikines), neurotransmitter‑inhibitor ("botox‑like") peptides, carrier peptides (e.g., copper peptides), and enzyme‑inhibitory peptides. Delivery constraints remain the key limitation — stratum corneum penetration varies dramatically by peptide size and vehicle, underscoring why outcomes can be inconsistent across products even with the same nominal peptide.
Exosomes: Cargo Transfer and Downstream Signalling
Exosomes are a subset of extracellular vesicles (EVs) involved in intercellular communication. Mechanistic plausibility is high at the concept level, but clinical translation is limited by: heterogeneity of exosome source (MSC/adipose/platelet/plant/bovine milk), isolation methods, potency assays, and delivery route.
Regulatory Warning: Exosomes
The US FDA has issued enforcement actions against companies marketing exosome products as unapproved drugs/biologics. In the UK, injected exosomes are considered medicinal products and lack UK marketing authorisation, making injectable use high legal/regulatory risk. This should be treated as a red‑flag area for institutional practice.
Sunekos and Jalupro: HA + Amino Acid/Peptide ECM Systems
Sunekos Performa is composed of low molecular weight HA plus a patented amino acid cluster (HY6AA) — it is not an exosome product despite frequent online confusion. Preclinical fibroblast screening shows formulation‑dependent ECM targeting for elastin and collagen IV.
Jalupro Super Hydro (HA + seven amino acids + three peptides) was evaluated in an open‑label observational study reporting: hydration +25.9% at 30 days post‑course, elasticity +29.2%, and collagen density +20.27%, with improvements maintained to 120 days.
Clinical Evidence: Efficacy, Effect Sizes, and Duration
Evidence Strength Hierarchy
- Highest: Topical tretinoin for photoageing wrinkles (SR/meta‑analysis of RCTs)
- Moderate: PRP (split‑face RCT); PN filler (active‑controlled RCT for crow's feet)
- Emerging: Exosomes (~20% wrinkle reduction across heterogeneous trials)
- Low‑to‑moderate: Topical peptides; Sunekos; Jalupro Super Hydro (observational)
Topical Tretinoin
A 2025 systematic review/meta‑analysis of RCTs (8 trials; 1,361 patients) found topical tretinoin significantly improved fine wrinkles (mean difference 0.412) and coarse wrinkles (mean difference 0.245) versus vehicle. Duration in included trials ranged from ~16 weeks to 2 years.
PRP (Split‑Face RCT)
In a split‑face randomised clinical trial (19 analysed participants), each participant received intradermal PRP on one cheek and saline on the other. At 6 months, participants rated PRP side significantly more improved for texture (2.00 vs 1.21; P=.02) and wrinkles (1.74 vs 1.21; P=.03) on a 5‑point improvement scale. Masked dermatologist ratings were "nominally but not significantly" superior for PRP.
Polynucleotide Filler (Crow's Feet RCT)
A phase III randomised, double‑blind matched‑pairs trial (PN filler vs HA filler) showed clinical photographic improvement ratio at 12 weeks was 95.7% for PN vs 94.3% for HA, meeting non‑inferiority criteria. Treatment‑emergent local adverse events were lower for PN (31.9%) than HA (48.6%).
PN‑HPT Priming + HA (Nasolabial Folds)
In a split‑face exploratory study (20 women), PN‑HPT priming then HA filler showed objective Antera 3D scores: wrinkles reduced from 36.1±1.76 to 27.6±2.47 at 6 weeks on the PN‑HPT side vs 35.3±1.39 to 33.7±2.25 on placebo side (p<0.05), with longer‑term texture and HA persistence advantages at 6 months.
Exosomes Meta‑Analysis
A 2026 systematic review/meta‑analysis reports facial wrinkle reduction averaging 20.2% (95% CI 15.3%–25.2%) and improvements in other outcomes (pigmentation, elasticity, texture, erythema) ranging ~14.7%–23.4%. Non‑standardised protocols and heterogeneity limit generalisability.
Jalupro Super Hydro (Observational)
The 2024 observational study reports hydration +25.9% at 30 days post‑course and +15.9% at 120 days; elasticity +29.2% and +20.7%; collagen density +20.27% and +16.71%, with minimal transient adverse effects.
Comparative Table for Clinicians
| Modality | Core Mechanism | Level of Evidence | Onset | Durability | Common AEs |
|---|---|---|---|---|---|
| Polynucleotides (PN/PDRN/PN‑HPT) | A2A receptor activation + nucleotide salvage support; pro‑angiogenic/ECM effects | Moderate (active‑controlled RCT for wrinkles; priming pilot RCT) | Weeks–Months | 6–12 months | Bruising/swelling; local AEs may be lower than HA |
| PRP | Platelet growth factors/cytokines; immune modulation/angiogenesis; highly protocol‑dependent | Moderate (split‑face RCT vs saline; meta‑analyses) | Weeks | ~3–12 months (variable) | Erythema, swelling, bruising; low allergy risk (autologous) |
| Retinoids (retinol/retinal/tretinoin) | RAR/RXR transcriptional changes; collagen/MMP/pigment pathways | High for tretinoin (RCT meta‑analysis) | 8–16 weeks | Maintained with continued use | Irritation, dermatitis; pregnancy avoidance |
| Topical Peptides | Neurotransmitter inhibition/enzyme inhibition; penetration limited by vehicle | Mixed (small trials; formulation issues) | 4–12 weeks | Requires continued use | Uncommon; efficacy varies |
| Exosomes | EV cargo transfer; signalling modulation; source and potency assays vary | Emerging; ~20% wrinkle reduction across heterogeneous trials | ~7 weeks | Unknown/variable | Mild transient erythema/oedema; safety reporting limited |
| Sunekos (HA + HY6AA) | HA hydration + amino‑acid driven ECM gene/protein responses (preclinical) | Limited controlled clinical data; stronger preclinical rationale | Weeks–Months | Practice‑dependent | Injection‑related bruising/swelling |
| Jalupro (AA + HA; Super Hydro) | HA hydration + amino acids/peptides; measured improvements in hydration/elasticity/collagen | Moderate‑emerging (open‑label objective measures) | ≥4 weeks | Improvements to 120 days post‑course | Transient ecchymosis/mild pain |
Safety, Adverse Events, and Contraindications
Across injectables, the dominant predictable risks remain procedure‑related (bruising, swelling, transient pain, vascular compromise depending on anatomy/technique) rather than molecule‑specific immunology — except where biologics are allogeneic or poorly regulated. The more "biologic" the product (PRP, exosomes), the more the clinician must control: sourcing, sterility, handling, and clear patient consent around uncertainty.
Polynucleotides
In the crow's feet RCT, treatment‑emergent local AEs: 31.9% for PN filler vs 48.6% for HA filler (statistically significant). No unexpected adverse device effects reported. Consider fish/salmonid allergies in counselling.
PRP
Low immunogenicity (autologous) but high procedural variability. RCT AEs: redness (n=18), swelling (n=16), bruising (n=14); none at 12 months. Exclusions: pregnancy, blood disorders, active skin disease, keloid history, immunosuppression.
Retinoids
Strongly evidence‑supported but predictable irritation is common; controlled ramp‑up and barrier strategies required. EU cosmetic retinol concentrations will be capped by regulation.
Exosomes
Meta‑analysis reports no serious/permanent AEs. However, regulatory non‑compliance is itself a patient safety risk. FDA warning letters, UK ATMP/medicines frameworks require marketing authorisation. Injectable use carries high legal/regulatory risk.
Patient Selection, Protocols, and Sequencing

Conceptual Sequencing for Clinicians
A useful mental model is "foundation → priming → procedural stimulation → maintenance":
Foundation
UV protection and barrier care plus retinoid where appropriate.
Priming
Transcriptional priming (retinoids), then injectable priming (polynucleotides/PN‑HPT) in dermal quality cases.
Procedural Stimulation
PRP, device‑based treatments, or HA/AA/peptide biorevitalisers depending on phenotype.
Maintenance
Lower‑intensity topicals + periodic booster sessions (6–12 monthly, phenotype‑dependent).
HSI Treatment Selection by Dominant Concern
| Dominant Concern | Recommended Track | Protocol Notes |
|---|---|---|
| Photoageing: fine rhytids / dyschromia | Topical retinoid programme (retinol/retinal → consider tretinoin) + sunscreen | Titrate from 2–3 nights/week; escalate as tolerated |
| Texture/roughness + accepts autologous variability | PRP course (standardised protocol + DEPA classification) | Monthly × 3 typical; reassess at 6 months |
| Crepey thin dermis / peri‑orbital quality | Polynucleotides / PN‑HPT (intradermal priming course) | Induction weeks apart; reassess at 12–16 weeks |
| Mild laxity / connective tissue support | Jalupro Super Hydro or AA/HA biorevitaliser (course‑based) | Monthly/fortnightly sessions; audit to 120 days |
| Under‑eye fine lines / early laxity | Sunekos (HA + HY6AA) course‑based | Document outcomes; limited controlled data |
| Patient requests "exosomes" | Governance check (topical only in UK risk frameworks) + informed consent | No injectable use without MHRA authorisation |
Practical Tips for Consistency
Standardisation beats novelty. For PRP, document preparation variables (spin protocol, platelet concentration targets, activation, injected volume), and use a classification approach such as DEPA to improve reproducibility and auditability.
Use objective measures where possible. Evidence studies frequently use imaging/biophysical tools (Antera 3D, DermaLab Combo, ultrasound). Replicating at least one objective measure in audits improves treatment calibration.
Priming logic: PN‑HPT priming data support the concept that "priming" before HA consolidation may enhance early wrinkle/texture improvements and potentially HA persistence. Use polynucleotides/PN‑HPT where the main outcome is dermal quality rather than volumisation, and add HA filler later only if structural support is still needed.
Cost, Consistency, and Variability
| Modality | Relative Cost | Variability | Notes |
|---|---|---|---|
| Retinoids | Lowest | Low | Ongoing topical programme; strong evidence makes cost‑effectiveness attractive |
| Polynucleotides | Moderate | Moderate | Device‑like product consistency; PN‑HPT priming as evidence‑based premium add‑on |
| PRP | Moderate–High | High | Time, consumables, staff; patient biology and device variability |
| Exosomes | High | High + Governance risk | Promising signals but standardisation and regulation remain limiting |
| Sunekos / Jalupro | Moderate–High | Low (product) / Mixed (evidence) | Brand value + course‑based care; Jalupro has quantified observational data |
Regulatory Status and Product Governance
Many "skin booster" injectables in Europe are marketed as CE‑marked medical devices (often Class III for injectable dermal products). Great Britain has transitional acceptance timelines for CE‑marked devices under the Medical Devices (Amendment) (Great Britain) Regulations 2023. This matters for procurement: CE acceptance remains relevant in GB, but governance must track certificate validity and transition schedules.
EU Commission Regulation (EU) 2024/996 restricts retinol/retinyl acetate/retinyl palmitate to 0.05% retinol equivalents in body lotions and 0.3% in other leave‑on/rinse‑off products. Clinicians should anticipate reformulations and counsel patients on the difference between OTC limits and prescription tretinoin evidence.
HSI Recommendations: Modality Positioning Summary
| Domain | Clinician Definition | Mechanistic Anchor | Evidence | HSI Recommendation |
|---|---|---|---|---|
| Polynucleotides | Purified DNA polymer fragments (often salmonid) delivered intradermally as gels/solutions | A2A receptor activation + nucleotide salvage support | RCT non‑inferior to HA filler for crow's feet; priming improves NLF outcomes | High‑value "priming" option; combine with retinoid foundation; audit outcomes |
| PRP | Autologous platelet concentrate injected intradermally/subdermally | Growth factor/cytokine milieu; highly prep‑dependent | Split‑face RCT shows patient‑rated improvements at 6 months | Offer with strict protocol standardisation and consent about variability |
| Retinoids | Topical vitamin A derivatives (OTC precursors; prescription RA) | RAR/RXR transcription; collagen/MMP/pigment pathways | Meta‑analysis: tretinoin improves fine/coarse wrinkles vs vehicle | Default first‑line priming; retinal first if tolerability priority |
| Topical Peptides | Diverse peptide actives in cosmetic vehicles | Signal/enzyme modulation; penetration‑limited | Mixed; meta‑analysis suggests benefits but heterogeneity limits generalisability | Adjunct only; avoid over‑promising; choose credible delivery data |
| Exosomes | Extracellular vesicles (variable sources) used topically or adjunct | Cargo transfer (RNAs/proteins/lipids) + uptake signalling | ~20% wrinkle reduction in meta‑analysis; heterogeneous | Governance gate; avoid injectables without authorisation; topical adjunct only |
| Sunekos | LMW HA + HY6AA amino acid cluster | HA hydration + ECM gene/protein responses (preclinical) | Preclinical support; clinical evidence variable/limited | Offer for peri‑orbital/crepey dermis in selected patients; document outcomes |
| Jalupro | HA + amino acids; Super Hydro adds peptides | HA hydration + amino acid/peptide support for dermis | Observational: hydration/elasticity/collagen improvements to 120 days | Consider for laxity/quality; build clinic audit out to 120 days |
Discussion and Conclusions
For a clinician audience, the most defensible, reproducible, and evidence‑anchored foundation remains retinoid therapy, especially tretinoin for photoageing‑associated wrinkles. A "retinal first" strategy is reasonable for tolerability and adherence — particularly in sensitive‑skin populations — while reserving tretinoin for patients who can tolerate escalation and desire maximal evidence‑based effect.
Injectables should be selected based on phenotype and governance. Polynucleotides/PN‑HPT merit a strong position for dermal quality priming. PRP should be offered as an autologous regenerative modality with explicit variability counselling. Jalupro (Super Hydro) can be positioned for laxity/quality cases. Sunekos can be positioned for under‑eye/thin dermis based on formulation rationale and preclinical evidence. Topical peptides are adjuncts rather than primary anti‑ageing engines.
Despite promising pooled outcomes in meta‑analysis (~20% wrinkle reduction), governance should prioritise regulatory clarity for exosomes and avoid injectable use under uncertain authorisation status. The clinician‑level question is less "which single booster is best?" and more "which combination yields consistent outcomes within a defensible governance framework."
Related HSI Training
References
- Systematic review/meta‑analysis of tretinoin RCTs for photoageing wrinkles, 2025. J Clin Med. MDPI
- PRP split‑face RCT for photoaged facial skin. JAMA Dermatology. JAMA Network
- PN filler vs HA filler phase III RCT (crow's feet). J Korean Med Sci. JKMS
- Jalupro Super Hydro open‑label observational study (hydration/elasticity/collagen density). Jalupro HCP resource.
- Sunekos fibroblast ECM gene/protein expression screening study. Preclinical data.
- Extracellular vesicles in intercellular communication. ScienceDirect. ScienceDirect
- Exosome systematic review/meta‑analysis of human trials, 2026. Aesthet Surg J. ASJ
- FDA Warning Letter: Kimera Labs Inc, 2023. FDA.gov
- PDRN wound‑healing review: A2A receptor activation and salvage pathway. Wound Repair Regen. SAGE Journals
- PDRN pharmacology — MDPI review. Pharmaceuticals
- PRP molecular mechanisms and clinical applications. MDPI IJMS. MDPI
- DEPA classification for PRP. BMJ Open Sport Exerc Med. BMJ
- RXR–RAR heterodimer structural biology. Nucleic Acids Res. NAR
- Retinoid conversion pathways. Int Cosmet Sci. Wiley
- Topical peptides systematic review/meta‑analysis. Front Med. Frontiers
- Acetyl hexapeptide‑8 permeation and efficacy review. MDPI IJMS. MDPI
- Exosomes — immunity and EV biology. Immunity. Cell Press
- PN‑HPT + HA split‑face exploratory study (NLFs). J Cosmet Dermatol. ResearchGate
- HA + amino acid fragments histological study. Springer. Springer Link
- EU Commission Regulation 2024/996 — retinol concentration limits. EUR‑Lex
- CE‑marked device acceptance timelines in Great Britain. GOV.UK
- UK ATMP regulation and licensing. GOV.UK
- PRP meta‑analysis 2026. ASJ Open Forum. ASJ Open Forum
